OUR TESTS
Breast Cancer Testing

Will my patient’s breast cancer return after surgery?

Will my patient’s breast cancer return after surgery?
The MammaPrint® test analyzes the 70 most important genes associated with breast cancer recurrence. Results are typically available in 6 days or less, MammaPrint enables quicker, more informed decisions on pre- and post-operative treatment and can easily be integrated into diagnostic workups.1

I see MammaPrint as a great tool which helps you to navigate through difficult decisions.”
— Andrea, a breast cancer survivor
I see MammaPrint as a great tool which helps you to navigate through difficult decisions.”
— Andrea, a breast cancer survivor
BREAST CANCER TREATMENT PLANNING
MammaPrint Test & Results
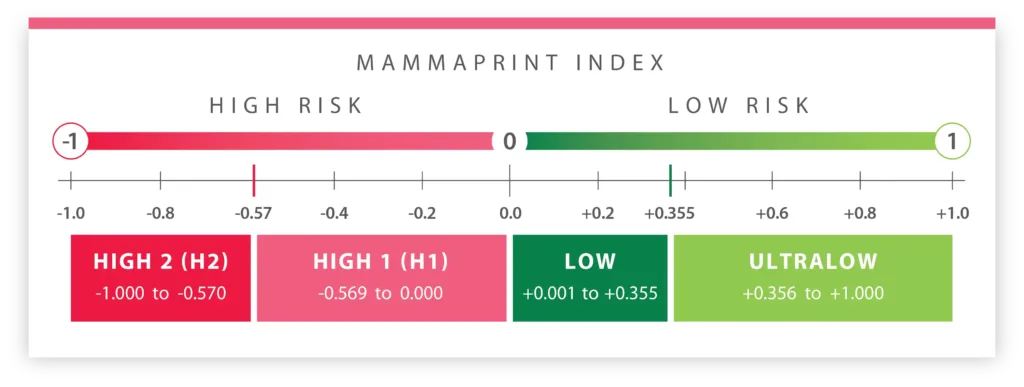
The MammaPrint Index
High Risk 1
Low Risk
UltraLow Risk
DEVELOPMENT
What Makes MammaPrint Different?
- Developed and validated on untreated patients – meaning, your result is reflective of as you are today, not based on completing a full 5 years of endocrine therapy like other tests may require for their result.
- Developed with genome-wide expression data using an unbiased data-driven approach without a preselection of genes to identify the 70 genes5
MammaPrint and BluePrint gene signatures represent and capture all 10 Hallmarks of Cancer and highlight underlying biological processes of MammaPrint extreme samples, which might guide treatment decisions as the signature captures the full spectrum of early breast cancers.6
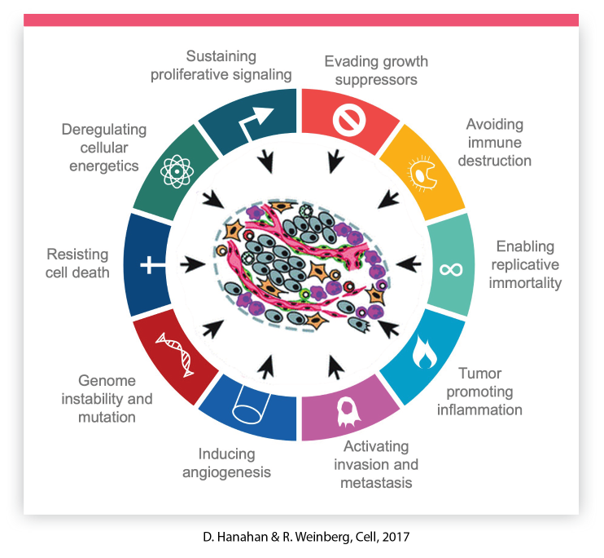
Definitive results for adjuvant chemotherapy planning.
LOW RISK PATIENTS HAVE A
1.3%
CHANCE OF RECURRENCE 2
The MINDACT trial determined these patients do not benefit from chemotherapy.3
HIGH RISK PATIENTS HAVE A
11.7%
CHANCE OF RECURRENCE 2
These patients were shown to have significantly better outcomes with chemotherapy.4
Nearly half of clinically high risk patients were reclassified as MammaPrint Low Risk and were able to forgo chemotherapy without compromising their outcomes.3
OUR PROOF
Implemented across the entire medical community.
MammaPrint is FDA-cleared for women of all ages. MammaPrint is also available via next generation sequencing on Illumina MiSeq platform. This approach has been CE marked allowing use in the European Union.
Latest MammaPrint Publications
- “Improved 3-Year IDFS With Anthracycline-Based Therapy for Patients With 70-Gene Signature High 2, Luminal B, HR+HER2- Early-Stage Breast Cancer”, O’Shaughnessy et al., SABCS 2025
- “MammaPrint Predicts Chemotherapy Benefit in HR+HER2- Early Breast Cancer: FLEX Registry Real-World Data”, Brufsky et al., JNCI Cancer Spectrum, August 2025
- “Identification of racial disparities across MammaPrint and BluePrint subtypes in HR+HER2- breast cancer”, Reid et al., npj Breast Cancer 2026
- “Utility of the 70-Gene MammaPrint Assay for Prediction of Benefit From Extended Letrozole Therapy in the NRG Oncology/NSABP B-42 Trial”, Rastogi et al., Journal of Clinical Oncology, July 2024
MammaPrint FAQ
1. Following the 2016 Publication of MINDACT, the 2017 ASCO guidelines were updated to indicate favorable results in patients with 1-3 positive lymph nodes. MammaPrint’s 510(k) FDA clearance includes breast cancer patients with Stage 1 or Stage II disease, with tumor size ≤ 5.0 cm and lymph node negative. (Agendia is committed to delivering results in less than 10 business days, and results are provided within 6 business days for the majority of cases)
2. FDA 510(k) clearance (K201902)
3. Cardoso, F., et al. N Engl J Med 2016;375:717-29.
4. Knauer, M., et al. Breast Cancer Res Treat. 2010 Apr;120(3):655-61.
5. Tian et al., Biomark Insights. 2010; 5: 129–138
6. Haan et al., Genes Chromosomes Cancer. 2022;61:148–160
* FDA Intended Use: the test is performed for breast cancer patients with Stage I or Stage II disease with tumor size ≤5.0cm and lymph node negative. The MammaPrint FFPE result is indicated for use by physicians as a prognostic marker only, along with other clinicopathological factors. Coverage criteria for majority of health plans (in the US) include early-stage breast cancer, tumor size less than or equal to 5.0cm (≤5.0cm), up to 3 positive lymph nodes and independent of receptor status as validated in the 6,693 patient MINDACT trial.
**FFPE = Formalin Fixed, Paraffin Embedded (a method for preserving clinical samples)
THE MAMMAPRINT + BLUEPRINT TEST SUITE
Analyze over a hundred relevant genes at once.
With the additional results of BluePrint, our molecular subtyping test, physicians gain an even broader genomic profile. From just one tumor block, you can interrogate over 150 genes containing information on a tumor’s level of aggression and potential for growth. These insights will serve as an essential resource for guiding your pre- and post-operative treatment strategies.






