PUBLICATION:
SABCS 2020
AUTHORS:
Ian E Krop, Lorenza Mittempergher, Joseph N Paulson, Fabrice Andre, Herve Bonnefo, Sherene Lo, Sibylle Lo1bl, Richard D Gelber’, Carmela Caballero, Debora Fumagall, RaJ1th Bhaskaran, Christa Dreezen, Rene Bernards’, Laura J van ‘t Veer and Martine J P1ccart
Background:
APHINITY is a phase Ill study (NCT01358877), which randomized 4805 patients with histologically centrally confirmed HER2+ early breast cancer (EBC) to standard adjuvant chemotherapy (C) and trastuzumab (T), plus either pertuzumab (P) or placebo’ for one year. With a median follow up of 45 months, the primary analysis showed significant invasive disease-free survival (iDFS) benefit at 3 years with a hazard ratio (HR) of 0.81 (95% Cl: 0.66-1.00; P = 0.045, 3-year IDFS percent 94.1 % vs 93.2%) for the addition of P to CIT1 . BluePrint (BP) is an 80-gene molecular subtyping test that classifies EBC into functional Basal, Luminal and HER2 BPsubtypes3 according to gene expression. Previous studies showed that this genomic test can reclassify HER2+ EBC (as determined by IHC/FISH) into Basal, Luminal or HER2 BPsubtypes4. Recently, it was revealed that a smaller proportion of breast tumours may display two functionally activated BP pathways (BP dual activated subtypes, of which one is usually less pronounced), whereas the majority has clearly only one activated functional BP pathway (BP single activated subtypes)5. We hypothesized that BluePrint could identify a subgroup of patients within the APHINITY population who derived further benefit from the addition of P to CIT.
Methods
Genomic results were obtained using RNA sequencing (RNAseq) data6 from a subset of APHINITY patients (N=969), which were derived from a 1023 unique patients nested casecontrol (NCC) set where event and matched controls were selected (1 iDFS event matched to 3 controls from the primary analysis database with 45 months median follow-up). Raw read counts were log2 transformed, followed by quantile normalization prior to genomic assessment. BP subtype scores were calculated in the same manner to the standard microarray diagnostic testing and calibrated based on a bridge analysis with matched microarray and RNAseq data. IDFS outcome based on genomic subtype and the treatment arm (P+CIT vs placebo + CIT) was analyzed. Results are reported descriptively with 95% Cls.
Results
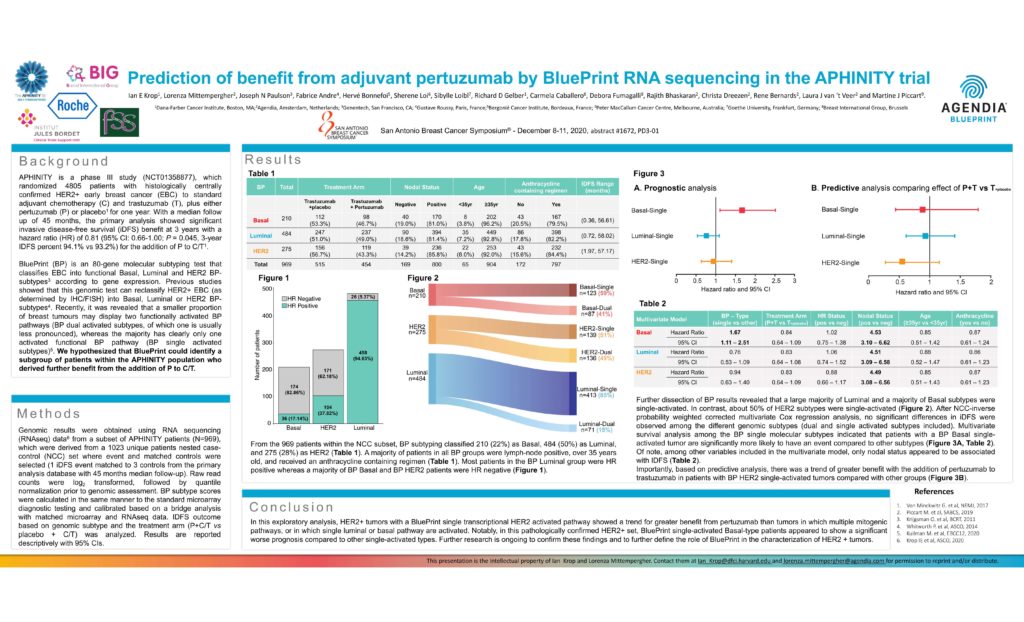
From the 969 patients within the NCC subset, BP subtyping classified 210 (22%) as Basal, 484 (50%) as Luminal, and 275 (28%) as HER2 (Table 1 ). A majority of patients in all BP groups were lymph-node positive, over 35 years old, and received an anthracycline containing regimen (Table 1 ). Most patients in the BP Luminal group were HR positive whereas a majority of BP Basal and BP HER2 patients were HR negative (Figure 1).
Conclusion
In this exploratory analysis, HER2+ tumors with a BluePrint single transcriptional HER2 activated pathway showed a trend for greater benefit from pertuzumab than tumors in which multiple mitogenic pathways, or in which single luminal or basal pathway are activated. Notably, in this pathologically confirmed HER2+ set, BluePrint single-activated Basal-type patients appeared to show a significant worse prognosis compared to other single-activated types. Further research is ongoing to confirm these findings and to further define the role of BluePrint in the characterization of HER2 + tumors.


