Title: MammaPrint Index predicts neoadjuvant chemosensitivity in patients with HR+HER2- early-stage breast cancer in the real-world evidence FLEX study
Publication: SABCS 2023, PO5-15-04
Authors
Joyce O’Shaughnessy, Lajos Pusztai, Cathy Graham, Pat Whitworth, Peter Beitsch, Cynthia Osborne, Rakhshanda Rahman, Emilee Russell,
Andrea Menicucci, William Audeh, FLEX Investigators’ Group
Background
Neoadjuvant chemotherapy (NCT) yields low pathologic complete response (pCR, ypT0/is ypN0) rates in hormone receptor positive (HR+), HER2- early-stage breast cancer (ESBC). Genomic signatures may predict chemosensitivity and treatment benefit better than receptor status based clinical subtyping. The 70-gene expression signature, MammaPrint (MP), classifies patients with ESBC as having a High or Low Risk of distant recurrence. In the NBRST and ISPY2 trials, further stratification of MammaPrint High Risk into High 1 (H1) or High 2 (H2) showed significantly higher pCR rates to NCT or targeted agents in MP H2 cancers compared to MP H1 tumors. We evaluated MammaPrint H1 and H2 status as a biomarker for neoadjuvant chemosensitivity in patients with HR+HER2- ESBC enrolled in the real-world evidence FLEX study.
Methods
FLEX (NCT03053193) is an ongoing prospective, observational trial that has currently enrolled 12,328 patients with ESBC who were tested with MammaPrint as standard of care, with or without molecular subtyping signature, BluePrint (BP), and consented to clinically annotated full genome data collection (data locked Feb. 2023). Patients with HR+HER2-, MP High Risk tumors who received NCT (majority received AC-T) and had pCR data available were included in this analysis (n = 214). Patients were stratified into MP H1 (index 0.000 to -0.569) and H2 (index -0.570 to -1.000) groups. BP classified MP High Risk tumors into Luminal B-, HER2-, or Basal-Type. Differences in clinical characteristics and pCR rates between H1 and H2 tumors was assessed by Chi-Squared test and two-sided proportional z-test, respectively. The association between MP H1 and H2, BP subtype, and pCR was assessed using logistic regression and was adjusted for age, race, grade, T stage, N stage, and NCT regimen.
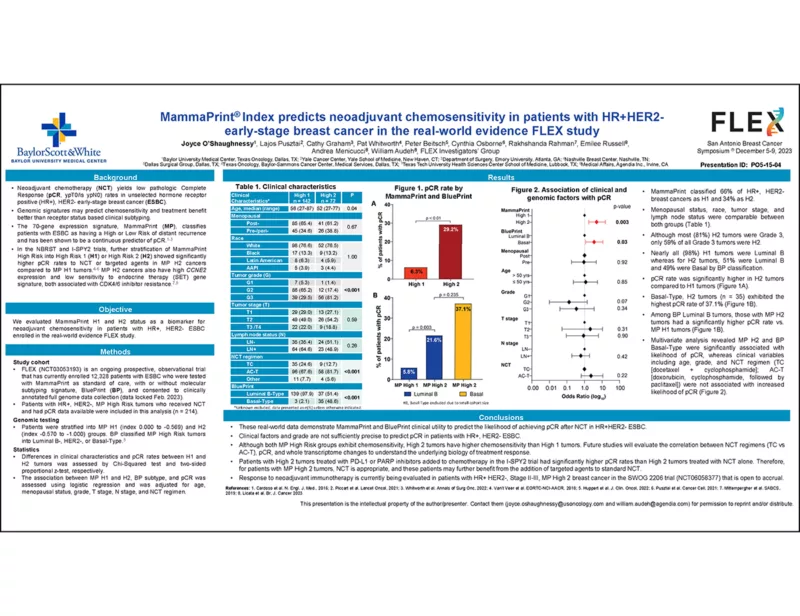
Results
MammaPrint classified 142 (66%) cancers as H1 and 72 (34%) as H2 tumors. Age, menopausal status, race, tumor stage, and lymph node status were comparable between both groups. Although most H2 tumors (78%) were Grade 3, only 59% of all Grade 3 tumors were H2. Nearly all (98%) H1 tumors were Luminal B whereas for H2 tumors, 51% were Luminal B and 49% were Basal by BP classification. pCR rate was significantly higher in H2 tumors (29.2%) compared to H1 tumors (6.3%, p < 0.01). Basal-Type, H2 tumors (n=35) exhibited the highest pCR rate of 37.1%. Among BP Luminal B tumors, those with MP H2 tumors had a significantly higher pCR rate (21.6%) vs. MP H1 tumors (5.8%, p = 0.003).
Multivariate analysis revealed MP H2 (OR=4.91, p=0.003) and BP Basal-Type (OR=3.54, p=0.03) were significantly associated with likelihood of pCR, whereas clinical variables were not associated with pCR.
Conclusions
These data demonstrate MammaPrint and BluePrint utility to predict the likelihood of achieving pCR after NCT in HR+HER2- ESBC. Although both MP High Risk groups exhibit chemosensitivity, High 2 tumors have higher chemosensitivity than High 1 tumors. MP High 2 status can be utilized to identify ER+ patients who are the most likely to experience pathologic downstaging and pCR after NCT. Also, patients with High 2 tumors treated with PD-L1 or PARP inhibitors in the ISPY2 trial exhibited significantly higher pCR rates than those observed for High 2 tumors treated with NCT alone. These data suggest that for patients with MP High 2 tumors, neoadjuvant use of chemotherapy is appropriate and these patients may further benefit from the addition of targeted agents to standard NCT.