Publication: ASCO 2023, Abstract #521
Authors
Peter D. Beitsch 1 , James V. Pellicane 2 , Lajos Pusztai 3 , Paul L. Baron 4 , Erin F. Cobain 5 , Mary K. Murray 6 , Andrew Ashikari 7 , Pond R. Kelemen 7 , Angela Marie Mislowsky 8 , Julie Barone 9 , Kenneth H. Cowan 10 , Rakhshanda Layeequr Rahman 11 , William C. Dooley 12 , Andrea Menicucci 13 , Christine Carruth 13 , M. William Audeh 13 , Pat W. Whitworth 14 , NBRST Investigators Group
1. Dallas Surgical Group, Dallas, TX; 2. Bon Secours Cancer Institute, Richmond, VA; 3. Yale University, New Haven, CT; 4. Lenox Hi ll Hospital, Northwell Health, New York, NY; 5. Michigan Medicine, Ann Arbor, MI; 6. Cleveland Clinic Akron General, Akron, OH; 7. Northwell Health, Sleepy Hollow, NY; 8. Tidelands Health, Coastal Carolina Breast Center, Murrells Inlet, SC; 9. Vail Health Shaw Regional Cancer Center, Edwards, CO ; 10. University of Nebraska Medical Center, Omaha, NE; 11. Texas Tech University Health Sciences Center School of Medicine, Lub bock, TX; 12. University of Oklahoma Health Sciences Center, Oklahoma City, OK; 13. Medical Affairs, Agendia, Inc., Irvine, CA; 14. Nashville Breast Center, Nashville, T N
Background & Objective
- Indications for neoadjuvant chemotherapy (NCT) in hormone receptor positive (HR+), HER2- breast cancer remain controversial (1).
- Pathological complete response (pCR) rates range from 0-18%, and breast conservation is possible in up to 60% of cases.
- Genomic signatures may predict pCR and benefit of NCT better than conventional subtypes.
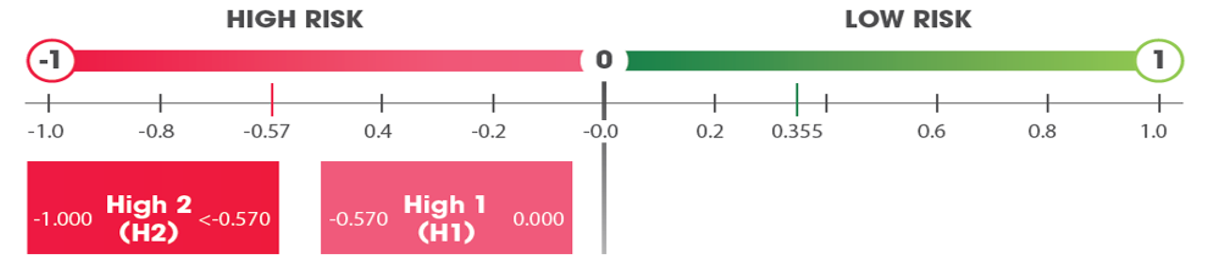
The 70-gene MammaPrint (MP) test classifies patients with early breast cancer as having a Low or High Risk of distant metastasis (2-3).
In the ISPY2 trial, further stratification of MP High Risk into High 1 or High 2 improved prediction of chemosensitivity, with significantly higher pCR rates in High 2 vs. High 1 tumors, particularly in response to immune therapy (4-6).
Objective: We evaluated the utility of High 1/High 2 risk as a biomarker for chemosensitivity and 5 year distant-metastasis free survival (DMFS) in NCT treated patients from the Neoadjuvant Breast Registry Symphony Trial (NBRST).
Methods
In the observational prospective NBRST study (6), a subset of patients with HR+HER2-, MammaPrint High Risk tumors who received NCT were included in this post-hoc analysis (n = 327).
Patient tumors were further stratified into MammaPrint High 1 or High 2 groups.
Statistical Analysis: Differences in clinical characteristics were assessed by Chi-Squared or Fisher’s exact test. Differences in pCR were assessed by two-sided proportional z-test. Differences in DMFS was evaluated by Kaplan Meier analysis and log-rank test. Median follow-up was 5.3 years.
Results
- pCR rates were significantly higher in patients with High 2 vs. High 1 tumors
- 198 patients with High 1 tumors; 129 patients with High 2 tumors
- pCR rates were also comparable between premenopausal and postmenopausal patients
- 5-year DMFS was significantly worse for patients with High 2 tumors compared with High 1 tumors
- Most events occurred early (< 3 years) in patients with High 2 tumors (75%) compared with High 1 tumors (48.8%)
- Patients with High 1 or High 2 tumors that achieved pCR had similar 5-year DMFS
- Of patients with residual disease, those with High 2 tumors had significantly worse outcomes than those with High 1 tumors
Conclusion:
- These data establish neoadjuvant treatment utility for MammaPrint, which predicts pCR in patients with HR+HER2- breast cancer.
- Although both MammaPrint High Risk groups exhibit chemosensitivity, High 2 risk tumors have higher chemosensitivity than High 1 tumors.
- In MP High Risk group, we did not see a difference in chemosensitivity based on menopausal status.
- These data highlight the critical importance of identifying patients with MammaPrint High 2 tumors early, in order to treat them neoadjuvantly and identify those that have residual disease to optimize treatment planning.
- Future studies should investigate whether addition of novel targeted therapies (i.e. CDK4/6 inhibitors or immune therapy) to standard NCT would enhance the pCR rates in these patient populations and improve outcome.
References and Acknowledgments:
1. Torrisi et al. CROH, 2021
2. Cardoso et al. NEJM, 2016
3. Piccart et al. Lancet Oncol, 2021
4. Van’t Veer et al. EORTC NCI AACR, 2018
5. Huppert et al. ASCO, 2022
6. Pusztai et al. Cancer Cell, 2021
7. Whitworth et al. Annals of Surg Onc , 2022


